|
10/29/2022 0 Comments Searchgui enzyme definition 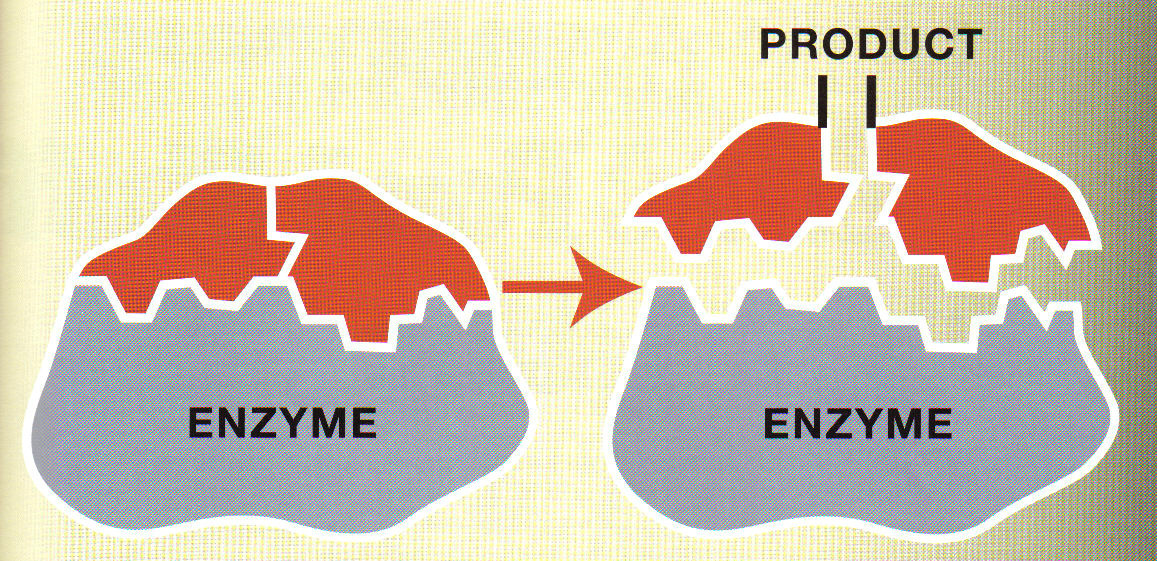
Non-vitamin coenzymes typically aid in chemical transfer for enzymes. Coenzyme A also initiates the citric acid cycle, resulting in the production of ATP. Fatty acids form the phospholipid bilayer that comprises the cell membrane, a feature necessary for life. 
First, it is responsible for initiating fatty acid production within cells. This coenzyme has several different functions. Additionally, this coenzyme is recognized as the most powerful biological antioxidant for protecting cells against harmful or damaging substances.Ĭoenzyme A, also known as acetyl-CoA, naturally derives from vitamin B5. A lack of NADH causes energy deficits in cells, resulting in widespread fatigue. For example, the electron transport chain can only begin with the delivery of electrons from NADH. This coenzyme primarily carries electrons for reactions and produces energy from food. In fact, it is considered the number one coenzyme in the human body because it is necessary for so many different things. NADH, often called coenzyme 1, has numerous functions. 
When NAD + gains electrons through a redox reaction, NADH is formed. If an organism has a NAD + deficiency, then mitochondria become less functional and provide less energy for cell functions. TAC results in other coenzymes, such as ATP. NAD + primarily transfers electrons needed for redox reactions, especially those involved in parts of the citric acid cycle (TAC). When NAD gains an electron, a high-energy coenzyme called NADH is formed. When NAD loses an electron, the low energy coenzyme called NAD + is formed. NAD is derived from vitamin B3 and functions as one of the most important coenzymes in a cell when turned into its two alternate forms. Two of the most important and widespread vitamin-derived coenzymes are nicotinamide adenine dinucleotide (NAD) and coenzyme A. Water-soluble vitamins, which include all B complex vitamins and vitamin C, lead to the production of coenzymes. If vitamin intake is too low, then an organism will not have the coenzymes needed to catalyze reactions. Many coenzymes, though not all, are vitamins or derived from vitamins. Instead, they are introduced to an organism in two ways: Vitamins Most organisms cannot produce coenzymes naturally in large enough quantities to be effective. These groups are often used in cellular respiration and photosynthesis. However, unlike coenzymes or cofactors, these groups bind very tightly or covalently to an enzyme to aid in catalyzing reactions. These can be organic vitamins, sugars, lipids, or inorganic metal ions. They must also be supplemented in the diet as most organisms do not naturally synthesize metal ions. Usually, cofactors are metal ions such as iron, zinc, cobalt, and copper that loosely bind to an enzyme’s active site. Unlike coenzymes, true cofactors are reusable non-protein molecules that do not contain carbon (inorganic). Most are vitamins, vitamin derivatives, or form from nucleotides. They bind loosely to an enzyme at the active site to help catalyze reactions. These are reusable non-protein molecules that contain carbon (organic). However, cofactors can be broken down into three subgroups based on chemical makeup and function: Coenzymes In general, all compounds that help enzymes are called cofactors. Types of EnzymesĬofactors are molecules that attach to an enzyme during chemical reactions. When an enzyme is denatured by extreme temperature or pH, the coenzyme can no longer attach to the active site. They attach to a portion of the active site on an enzyme, which enables the catalyzed reaction to occur. Coenzymes, like enzymes, can be reused and recycled without changing reaction rate or effectiveness. Active enzymes change substrates into the products an organism needs to carry out essential functions, whether chemical or physiological. When an enzyme gains a coenzyme, it then becomes a holoenzyme, or active enzyme. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
